 
In the 5th Step➞ atoms CAN be divided further (3 subatomic particles: protons, electrons, and neutrons). In the 2nd Step ➞ atoms of a given element are NOT always identical (isotopes). Today, some 215 years after John Dalton proposed his Atomic Theory of Matter in 1808, there are just two modifications that must be made: Dalton’s Atomic Theory Modifications: = The Modifications of Dalton’s Atomic Theory What was John Dalton’s atomic model called Based on all his observations, Dalton proposed his model of an atom. #5 - Atoms cannot be divided any further. Daltons atomic theory proposed that all matter was composed of atoms, indivisible and indestructible building blocks. What did John Dalton accomplish John Dalton (1766-1844) was an English chemist, physicist, and meteorologist, best known for introducing the atomic theory into chemistry and for his work on human optics.But the atoms themselves are not changed during the process. #4 - Chemical reactions involve the re-organization of atoms.A given compound always has the same relative numbers and types of atoms. According to Democrituss atomic theory, all matter is made up of very small particles and he called these particles atoms. #3 - Compounds are formed when atoms of different elements are combined.The atoms of different elements are different. When elements were excited by an electrical current. #2 - The atoms of a given element are identical. Thomson (18561940) disproved Daltons idea that atoms are indivisible.#1 - Each element is composed of tiny indestructible particles called atoms.The five postulates below summarize Dalton’s Atomic Theory… General Chemistry Online! Dalton's atomic theoryĬomments & questions to Revised 02/23/18.Dalton’s Atomic Theory of Matter, and his 5 Postulates Dalton’s Atomic Theory and 5 Postulates : Learning check Take a quiz on Dalton's Atomic Theory : Atoms in compounds An "invisible hand" fixes atom ratios in compounds : Dalton's assumptions Five steps forward and one step back. 
But the core concepts of the theory (that chemical reactions can be explained by the union and separation of atoms, and that these atoms have characteristic properties) are foundations of modern physical science. Some of the details of Dalton's original atomic theory are now known to be incorrect. He was the first to associate the ancient idea of atoms with stoichiometry. He realized the vital theoretical connection between atomic weights and weight relations in chemical reactions. Thus a train of investigation was laid for determining the number and weight of all chemical elementary particles which enter into any sort of combination one with another."ĭalton's exceptional gift for recognizing and interpreting patterns in experimental data lead him from a problem in meteorology to the idea of atoms as fundamental constituents of matter. ".it became an object to determine the relative sizes and weights, together with the relative numbers of atoms entering into such combinations. John Daltons atomic theory was generally accepted because it explained the laws of conservation of mass, definite proportions, multiple proportions, and other. 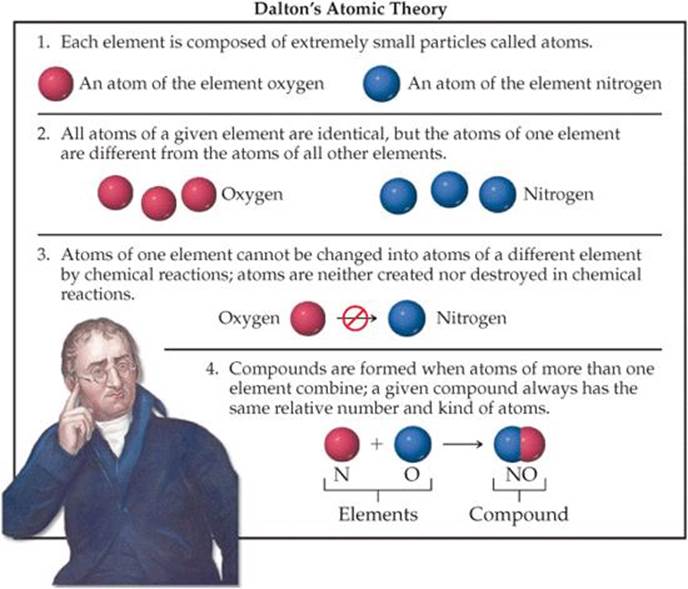
While trying to explain the results of those experiments, Dalton developed the hypothesis that the sizes of the particles making up different gases must be different. He performed a series of experiments on mixtures of gases to determine what effect properties of the individual gases had on the properties of the mixture as a whole. If the water and air were composed of discrete particles, Dalton reasoned, evaporation might be viewed as a mixing of water particles with air particles. He wondered how water and air could occupy the same space at the same time, when obviously solid bodies can't. According to him, all elements are made of atoms and the purity of an element is attributed to the presence of similar atoms. In the course of his studies on meteorology, Dalton concluded that evaporated water exists in air as an independent gas. John Dalton, the father of modern atomic theory, developed the atomic theory in the early nineteenth century. Useful atomic theory of matter around 1803. John Dalton (1766-1844) developed the first We might as well attempt to introduce a new planet into the solar system, or to annihilate one already in existence, as to create or destroy a particle of hydrogen. According to John Dalton, all matter, whether an element, a compound or a mixture, is composed of tiny particles called atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
